I'm looking for information about
Can't find what you're looking for? You may need to login to see more documents
Suggested parameters and sets of instructions outlining best practices and standards for accomplishing specific duties.
This Copy Was Generated On: October 1, 2025
Seeking Reconsent from Research Participants
IRBMED
| Approval Date:
July 29, 2020 2:00 pm
Informed consent is an ongoing, interactive process throughout a participant’s study involvement, rather than a one-time information session. In addition to encouraging subjects to ask questions and raise concerns, it may become necessary to seek renewed informed consent from a participant when aspects of a research study are altered during their participation. The circumstances that may warrant participants’ reconsent, as well as the available methods employed to obtain it, will be the subject of this guidance.
Substantive Changes
Any change that may change a participant’s willingness to continue in the study requires reconsent. However, it is not changes that modify the risk/benefit ratio that only warrant reconsent. Federal regulations from OHRP (45 CFR 46.116 (b) (5)) and FDA (21 CFR 50.25 (b) (5)) state that, when appropriate, the informed consent document must include a statement that “significant new findings developed during the course of the research which may relate to the participant’s willingness to continue participation will be provided to the participant.”
Examples:
- increasing the risks, for instance the identification of new risks or a change in the magnitude of known or
suspected risks - decreasing the potential for benefit
- availability of new alternative treatments
- FDA approval of an investigational drug (so that the drug is available outside the research context)
- adding study procedures; and
- modifying or removing study procedures when this can affect risk or potential
benefit.
Please note: Changes to study procedures that do not affect risk or potential benefit still warrant a reconsent process if already enrolled participants need to agree to those additional procedures.
Changes to Previously Approved Application via an Amendment
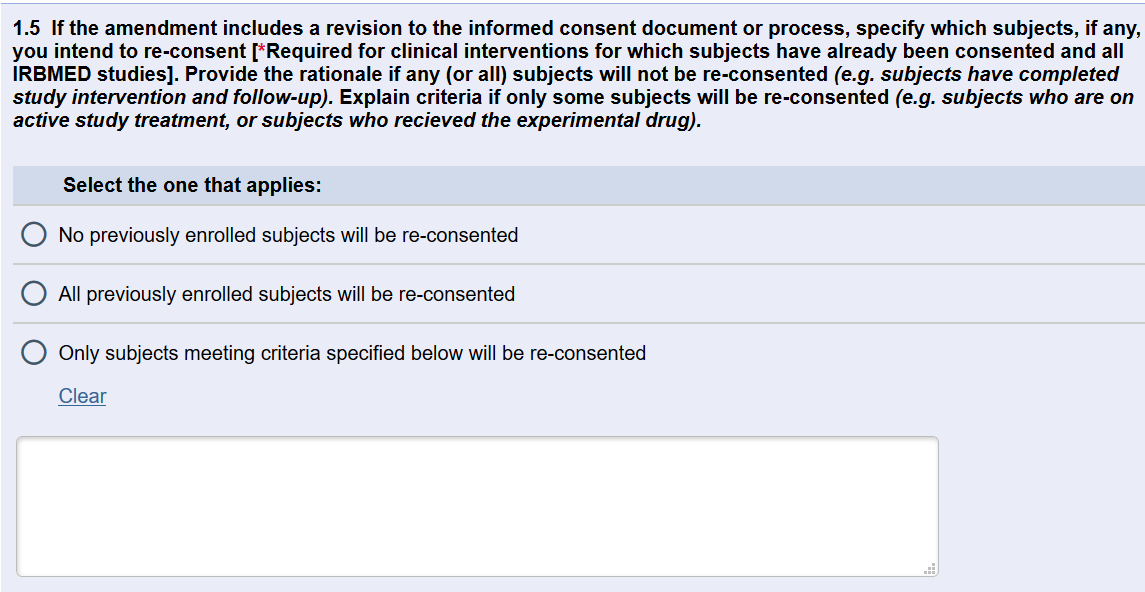
The eResearch Regulatory Management application asks for each Amendment submission (“Edit Amendment Forms,” also known as “Amendment Cover Sheet”) whether participants’ reconsent will be sought as a result of the changes being made. In 1.3, a checkmark should indicate that section 10 (Informed Consent) has been updated; then 1.5 must be completed.
When Children Turn 18
When minors turn age 18, and if they are continuing as research participants, they must provide consent using the adult informed consent document. OHRP Research with Children FAQ states that “unless the Institutional Review Board (IRB) determines that the requirements for obtaining informed consent can be waived, the investigators should seek and obtain the legally effective informed consent, as described in 45 CFR 46.116, for the now-adult participants for any ongoing interactions or interventions with the participants.”
This includes ongoing use of data or biospecimens collected for a research repository.
The eResearch Regulatory Management application asks for each study enrolling minors (section 10.2 Informed Assent) a participant’s status as a child may change during the study (e.g., reaching the age of majority), and what the plans are for seeking the individual’s consent.

Intermittent Cognitive/Decisional Impairment
When study participants who were temporarily cognitively impaired regain consent capacity, they should be undergo a full consent process. If participants who regain consent capacity decide that they do not wish to continue to participate in the study, their decision must be respected. This would involve withdrawing the participant and/or
their data/specimens. This situation arises in research conducted in critical or emergency situations, where the initial consent may have been provided by a legally authorized representative (LAR).
The eResearch Regulatory Management application asks for each study enrolling adults with impaired cognition or decision-making capacity (sections 10.1 Informed Consent, and 37) whether the cognitive capacity of the subjects is expected to change significantly during the study, and what the plans are for seeking such individuals’ consent.

Improperly Obtained Consent
Situations where participant may need to be asked to reconsent:
- When informed consent was obtained improperly. For example, if the person who obtained consent was not authorized to do so. (e.g. not listed in section 1.3 of the eResearch application as study personnel).
- When the wrong version of the Informed Consent Document has been used and the consent form varies by more than the version date (e.g. the content is not the same).
- The study team utilized version of the consent document that was not watermarked (approved) by IRBMED.
Also submit a “Protocol deviation” ORIO (Other Related Information or Occurrence)
Permissible Methods for Seeking Reconsent
Study teams may use various methods of reconsent:
- A fully revised consent document
- Written consent document addendum
- A telephonic re-consent process for specific study activity
Examples of When Reconsent May Not Be Necessary
Sometimes notifying participants of changes made is enough and a formal reconsent process isn’t necessary.
Please note: Final decisions for individual studies are up to the board reviewer(s).
Examples:
- Key study team member changes without contact information changing
- The wrong version of the informed consent document was used, but the only difference is the version date
- Formatting or typographical errors in the informed consent document that do not change the meaning, understandability, or contact information
- Removing questions on a repeated survey that do not change the scope or sensitivity of the survey
- Decreasing the number or frequency of repeated procedures, if this does not affect data needed to ensure participant safety
Questions?
Contact us at [email protected] or 734-763-4768 / (Fax 734-763-1234)
2800 Plymouth Road, Building 520, Room 3214, Ann Arbor, MI 48109-2800
A list of IRBMED staff is available in the Personnel Directory, or view the list of Regulatory Teams.
Edited By: [email protected]
Last Updated: September 17, 2021 10:30 PM

